Histopathological investigation of the effect of levofloxacin and cephalexin derivative antibiotics on bone healing in experimentally induced fractures in rat tibias.
Abstract
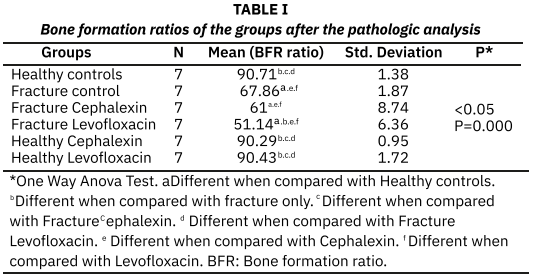
This study compared the effects of systemic Levofloxacin and Cephalexin on bone healing in a rat tibial fracture model. The subjects were divided into six groups (n=7 each): healthy control, fracture control, healthy Levofloxacin, healthy Cephalexin, fractured Levofloxacin, and fractured Cephalexin. In total, 42 Sprague–Dawley rats were used. In the fractured groups, a transverse fracture was created in the right tibial diaphysis, and the bone fragments were stabilized with Kirschner wire. No surgical intervention was performed in the healthy control and healthy antibiotic groups. The antibiotic groups received systemic Levofloxacin (25 mg/kg) or Cephalexin (20 mg/kg) three times a week for 4 weeks, starting on the same day as the fractured subjects. At the end of the experimental process, all animals were euthanised. After the decalcification process, the obtained samples were evaluated histopathologically, and bone filling ratio percentages were analyzed using histomorphometric methods. In the statistical analysis, differences between groups were examined using one- way analysis of variance. In cases where the variances were not homogeneous, pairwise comparisons were performed using the Games–Howell post-hoc test. Bone formation ratios (%) showed significant differences between the groups (P < 0.05). In this study, bone formation rates were found to be 90.71 in the healthy control group, 67.86 in the fracture control group, 61 in the fracture cefelaxin group, 51.14 in the fracture levofloxacin group, 90.29 in the healthy cefelaxin group, and 90.43 in the healthy levofloxacin group. While there was no difference in bone formation among the healthy groups, it was determined that bone formation was reduced in the fractured groups. Among the fractured groups, bone formation in the group treated with Levofloxacin was found to be significantly lower compared to the groups treated with only fracture and Cephalexin (P < 0.05). Considering the results of this study, it is thought that levofloxacin, in particular, has a bone formation-suppressing effect.
Downloads
References
Ielo I, Calabrese G, De Luca G, Conoci S. Recent Advances in Hydroxyapatite-Based Biocomposites for Bone Tissue Regeneration in Orthopedics. Int. J. Mol. Sci. [Internet]. 2022; 23(17):9721. doi: https://doi.org/g8rzn6 DOI: https://doi.org/10.3390/ijms23179721
Omi M, Mishina Y. Roles of osteoclasts in alveolar bone remodeling. Genesis. [Internet]. 2022; 60(8-9):e23490. doi: https://doi.org/gtz76m DOI: https://doi.org/10.1002/dvg.23490
Sun J, Xie W, Wu Y, Li Z, Li Y. Accelerated bone healing via electrical stimulation. Adv Sci. [Internet]. 2025; 12(24):2404190. doi: https://doi.org/g8694w DOI: https://doi.org/10.1002/advs.202404190
Fan S, Sun X, Su C, Xue Y, Song X, Deng R. Macrophages-bone marrow mesenchymal stem cells crosstalk in bone healing. Front. Cell Dev. Biol. [Internet]. 2023; 11:1193765. doi: https://doi.org/17, 18, 19] DOI: https://doi.org/10.3389/fcell.2023.1193765
ElHawary H, Baradaran A, Abi-Rafeh J, Vorstenbosch J, Xu L, Efanov JI. Bone healing and inflammation: principles of fracture and repair. Semin. Plast. Surg. 2021; 35(3):198-203. doi: https://doi.org/q47h DOI: https://doi.org/10.1055/s-0041-1732334
Maruyama M, Rhee C, Utsunomiya T, Zhang N, Ueno M, Yao Z, Goodman SB. Modulation of the inflammatory response and bone healing. Front. Endocrinol. [Internet]. 2020; 11:386. doi: https://doi.org/gjtsz6 DOI: https://doi.org/10.3389/fendo.2020.00386
Moriarty TF, Metsemakers WJ, Morgenstern M, Hofstee MI, Vallejo-Diaz A, Cassat JE, Wildemann B, Depypere M, Schwarz EM, Richards RG. Fracture-related infection. Nat. Rev. Dis. Primers. [Internet]. 2022; 8(1):67. doi: https://doi.org/mq9t DOI: https://doi.org/10.1038/s41572-022-00396-0
Shu LZ, Zhang XL, Ding YD, Lin H. From inflammation to bone formation: the intricate role of neutrophils in skeletal muscle injury and traumatic heterotopic ossification. Exp. Mol. Med. [Internet]. 2024; 56(7):1523-1530. doi: https://doi.org/g8xfdh DOI: https://doi.org/10.1038/s12276-024-01270-7
Camarena A, Kang L, Mirando AJ, Augustine E, McMillian NS, Stinson NC, Agarwal SM, Becker ML, Hilton MJ, Fernandez-Moure JS. Platelet-rich plasma enhances rib fracture strength and callus formation in vivo. J. Trauma Acute Care Surg. [Internet]. 2024; 97(6):884-890. doi: https://doi.org/qmx3 DOI: https://doi.org/10.1097/TA.0000000000004441
Trompet D, Melis S, Chagin AS, Maes C. Skeletal stem and progenitor cells in bone development and repair. J. Bone Miner. Res. [Internet]. 2024; 39(6):633-654. doi: https://doi.org/gt2h8d DOI: https://doi.org/10.1093/jbmr/zjae069
Hente RW, Perren SM. Tissue deformation controlling fracture healing. J. Biomech. [Internet]. 2021; 125:110576. doi: https://doi.org/qmx4 DOI: https://doi.org/10.1016/j.jbiomech.2021.110576
Kondi S, Gowda SR. Principles of bone healing. Surgery. [Internet]. 2023; 41(10):625-631. doi: https://doi.org/gmx5 DOI: https://doi.org/10.1016/j.mpsur.2023.08.002
Can UK, Tanrisever M, Ozcan EC, Dogan D, Istek O, Donmezer T, Karabulut B. Dundar S. Histopathological examination of healing in bone defects in intermittent fasting: An experimental study. Rev. Cient. FCV-LUZ. [Internet]. 2026; 36(1): e361832 doi: https://doi.org/q47k DOI: https://doi.org/10.52973/rcfcv-e361832
Flores MJ, Brown KE, Morshed S, Shearer DW. Evidence for Local Antibiotics in the Prevention of Infection in Orthopaedic Trauma. J. Clin. Med. [Internet]. 2022; 11(24):7461. doi: https://doi.org/q47n DOI: https://doi.org/10.3390/jcm11247461
Cook MA, Wright GD. The past, present, and future of antibiotics. Sci. Transl. Med. [Internet]. 2022; 14(657):e7793. doi: https://doi.org/gsmqxp DOI: https://doi.org/10.1126/scitranslmed.abo7793
Dawoud BES, Kent S, Henry A, Wareing J, Chaudry H, Kyzas P; MTReC; Kyzas P. Use of antibiotics in traumatic mandibular fractures: a systematic review and meta-analysis. Br. J. Oral Maxillofac. Surg. [Internet]. 2021; 59(10):1140-1147. doi: https://doi.org/g7jkwq DOI: https://doi.org/10.1016/j.bjoms.2021.01.018
Stockwell E, Rinehart K, Boes E, Pietrok A, Hewlett A, Hartman C, Streubel P. Outcomes of Orthopaedic Infections in Recreational Intravenous Drug Users Requiring Long-term Antibiotic Treatment. J. Am. Acad. Orthop. Surg. [Internet]. 2022 6(6):e22.00108.doi: https://doi.org/q47p DOI: https://doi.org/10.5435/JAAOSGlobal-D-22-00108
Yang Z, Meng H, Li J, Du P, Lv H, Zhao K, Zhang J, Li M, Jin Z, Peng Z, Ye D, Ding K, Song Z, Wang J, Xing X, Zhu Y, Zhang Y, Chen W. Association between the generation of cephalosporins for perioperative prophylaxis and postoperative surgical site infections in open fractures: a prospective cohort study. Int. J. Surg. [Internet]. 2025; 111(6):3810-3820. doi: https://doi.org/q47q DOI: https://doi.org/10.1097/JS9.0000000000002371
Uslu M, Yılmaz B, Mraja HM, Daşcı MF, Yaprak-Saraç E, Küçükyıldırım BO, Güleç MA, Yüksel S. The effects of cephalexin on fracture healing in a rat femur fracture model. Joint. Dis. Relat. Surg. [Internet]. 2023; 34(2):413-424. doi: https://doi.org/q47r DOI: https://doi.org/10.52312/jdrs.2023.994
Baggio D, Ananda-Rajah MR. Fluoroquinolone antibiotics and adverse events. Aust. Prescr. [Internet]. 2021; 44(5):161-164. doi: https://doi.org/jmh4 DOI: https://doi.org/10.18773/austprescr.2021.035
Brar RK, Jyoti U, Patil RK, Patil HC. Fluoroquinolone antibiotics: An overview. Adesh Univ. J. Med. Sci. Res. [Internet]. 2020; 2(1):26-30. doi: https://doi.org/q478 DOI: https://doi.org/10.25259/AUJMSR_12_2020
Can UK, Tanrisever M, Dagtekin B, Pak P, Ozcan EC, Istek O, Dundar S. Evaluation of the effect of Levofloxacin and Cefalexin derivative antibiotics on Implant Osseointegration in rat tibia. Rev. Cient. FCV-LUZ, [Internet]. 2025; 35(3):e35679. doi: https://doi.org/q479 DOI: https://doi.org/10.52973/rcfcv-e35679
Sheen JR, Mabrouk A, Garla VV. Fracture Healing Overview. In: StatPearls [Internet]. 2023 Apr 8. Treasure Island, Florida, USA: StatPearls Publishing; [Internet]. 2026 [cited 22 Dec 2025]. Available in: https://goo.su/odZnKa
Pajarinen J, Lin T, Gibon E, Kohno Y, Maruyama M, Nathan K, Lu L, Yao Z, Goodman SB. Mesenchymal stem cell-macrophage crosstalk and bone healing. Biomaterials. [Internet]. 2019; 196:80-89. doi: https://doi.org/gh4vp4 DOI: https://doi.org/10.1016/j.biomaterials.2017.12.025
Tanrisever M, Tekin B, Can UK, Istek O, Ozcan EC, Ozercan IH, Gelic T, Dundar S. The Effect of Local Melatonin Application on Bone Fracture Healing in Rat Tibias. Medicina. [Internet]. 2025; 61(1):146. doi: https://doi.org/gmzf DOI: https://doi.org/10.3390/medicina61010146
Krischak G, Augat P, Blakytny R, Claes L, Kinzl L, Beck A. The non-steroidal anti-inflammatory drug diclofenac reduces appearance of osteoblasts in bone defect healing in rats. Arch. Orthop. Trauma Surg. [Internet]. 2007; 127(6):453-458. doi: https://doi.org/bbkcw3 DOI: https://doi.org/10.1007/s00402-007-0288-9
Özbay H, Yüksel S, Güleç MA, Atçı T, Küçükyıldırım BO, Çay T. The effect of different irrigation solutions on fracture healing in a rat femur fracture model. Joint Dis. Relat. Surg. [Internet]. 2021; 32(1):144-151. doi: https://doi.org/q48g DOI: https://doi.org/10.5606/ehc.2021.77358
Zhang F, Liu F, Yu S, Zhang G, Li J, Sun X. Protective effect of curcumin on bone trauma in a rat model via expansion of myeloid derived suppressor cells. Med. Sci. Monit. [Internet]. 2020; 26:e924724. doi: https://doi.org/gq4q6k DOI: https://doi.org/10.12659/MSM.924724
Güven N, Özkan S, Türközü T, Koç S, Keleş ÖF, Yener Z, Karasu A. The effect of theranekron on femur fracture healing in an experimental rat model. Joint Dis. Relat. Surg. [Internet]. 2022; 33(2):374-384. doi: https://doi.org/g48j DOI: https://doi.org/10.52312/jdrs.2022.640
Huddleston P, Steckelberg J, Hanssen A, Rouse M, Bolander M, Patel R. Ciprofloxacin inhibition of experimental fracture-healing. J. Bone Joint Surg. Am. [Internet]. 2000; 82(2):161-173. doi: https://doi.org/psz2 DOI: https://doi.org/10.2106/00004623-200002000-00002
Perry AC, Prpa B, Rouse MS, Piper KE, Hanssen AD, Steckelberg JM, Patel R. Levofloxacin and trovafloxacin inhibition of experimental fracture-healing. Clin. Orthop. Relat. Res. [Internet]. 2003; 414:95-100. doi: https://doi.org/fpndfm DOI: https://doi.org/10.1097/01.blo.0000087322.60612.14
Golestani S, Golestaneh A, Gohari AA. Comparative effects of systemic administration of levofloxacin and cephalexin on fracture healing in rats. J. Korean Assoc. Oral Maxillofac. Surg. [Internet]. 2022; 48(2):94-100. doi: https://doi.org/pszz DOI: https://doi.org/10.5125/jkaoms.2022.48.2.94
Durmaz B, Gunes N, Koparal M, Gul M, Dundar S, Bingul MB. Investigation of the effects of quercetin and xenograft on the healing of bone defects: An experimental study. J. Oral Biol. Craniofac. Res. [Internet]. 2023; 13(1):22-27. doi: https://doi.org/pmp9 DOI: https://doi.org/10.1016/j.jobcr.2022.10.008