Efficacy of Punica granatum L. and Matricaria chamomilla L. herbal extracts on wound healing
Abstract
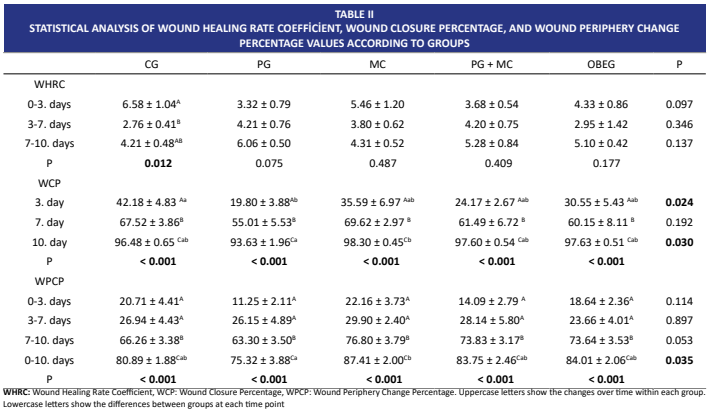
Dermal injuries, which account for a significant portion of healthcare expenditures globally, occur in various forms due to the influence of various etiological factors, primarily trauma. For all these reasons, wound healing is among the most researched topics worldwide. This study aimed to investigate the effects of ointments obtained from Punica granatum L. (pomegranate) and Matricaria chamomilla L. (chamomile) extracts on secondary wound healing. 42 Sprague Dawley rats were used in the study. In the study, a total of six experimental groups were formed, each consisting of 7 rats: control group, healthy control group, Punica granatum treatment group, Matricaria chamomilla treatment group, Punica granatum and Matricaria chamomilla combination group (Punica granatum treatment + Matricaria chamomilla treatment), and the group receiving an application containing olive oil, beeswax, and natural vitamin E. An excisional wound model was created using a biopsy punch on the back of the subjects in all groups except the healthy control group. While ointments prepared from herbal extracts were applied to the rats in the Punica granatum treatment, Punica granatum treatment + Matricaria chamomilla treatment, and Matricaria chamomilla treatment groups, rats in the control group received 0.9 % NaCl, and rats in the olive oil, beeswax, and natural vitamin E group were treated with a mixture of olive oil, beeswax, and vitamin E without any herbal extracts. At the end of the study, it was determined that the Matricaria chamomilla treatment group had the highest mean in terms of wound closure percentage and wound perimeter change percentage, while the Punica granatum treatment group had the lowest mean (P < 0.05). Although the best groups in terms of histopathological scoring were the Punica granatum treatment and Matricaria chamomilla treatment groups, no statistical difference was observed between the groups (P > 0.05). In terms of oxidative stress parameters (except for superoxide dismutase), the treatment groups were statistically significantly closer to the healthy control group than to the control group. Considering all evaluation criteria, it was concluded that Matricaria chamomilla plant was more effective on wound healing than Punica granatum plant and was a more suitable option for clinical use.
Downloads
References
Baktır G. Wound repair and experimental wound models. Experimed. [Internet]. 2019; 9(3):130-137. doi: https://doi.org/gp3xn4 DOI: https://doi.org/10.26650/experimed.2019.19023
Polat E, Karademir K, Cevik HN, Başar C, Kaya E, Ozoner O, Pekince-Ozoner M. Effect of Microcurrent Electrical Therapy on Healing of Excisional Skin Wounds. Rev. MVZ Cordoba. [Internet]. 2025; 30(2):e3681. doi: https://doi.org/qsm3 DOI: https://doi.org/10.21897/rmvz.3681
Polat E. Infected Chronic and İnjured Dog Henna (Lawsonia inermis) Therapeutic Use of Plants. Harran Univ. Vet. Fak. Derg. [Internet]. 2014 [cited 13 Sept 2025]; 3(2):93-97. Available in: https://goo.su/pUAzft
Parsak CK, Sakman G, Çelik U. Wound Healing, Wound Care and Its Complications. Arşiv. Kainak Tarama Derg. [Internet]. 2007 [cited 12 Sept 2025]; 16(2):145-159. Available in: https://goo.su/xPadG
Niknam S, Tofighi Z, Faramarzi MA, Abdollahifar MA, Sajadi E, Dinarvand R, Toliyat T. Polyherbal Combination for Wound Healing: Matricaria chamomilla L. and Punica granatum L. Daru. J. Pharm. Sci. [Internet]. 2021; 29(1):133-145. doi: https://doi.org/gjxh52 DOI: https://doi.org/10.1007/s40199-021-00392-x
Murthy KNC, Reddy VK, Veigas JM, Murthy UD. Study on Wound Healing Activity of Punica granatum Peel. J. Med. Food. [Internet]. 2004; 7(2):256-259. doi: https://doi.org/cvx47w DOI: https://doi.org/10.1089/1096620041224111
Sucharitha PAM, Poojitha M, Sravani B, Ganga PSL, Sai- Teja K, Chandana M, Kalpana S, Kumar SN. Preparation and Evaluation of Wound Healing Herbal Ointment. Int. J. Curr. Innov. Adv. Res. [Internet]. 2025; 8(2): 1-5. doi: https://doi.org/qsm4 DOI: https://doi.org/10.47957/ijciar.v8i2.196
Cukjati D, Rebersek S, Miklavcic C. A reliable method of determining wound healing rate. Med. Biol. Eng. Comput. [Internet]. 2001; 39:263-271. doi: https://doi.org/fg7rhx DOI: https://doi.org/10.1007/BF02344811
Greenhalgh DG, Sprugel KH, Murray MJ, Ross R. PDGF and FGF Stimulated Healing in the Genetically Diabetic Mouse. Am. J. Pathol. [Internet]. 1990 [cited 30 Jun 2025]; 136(6):1235-1246. Available in: https://goo.su/ZKcnSZ
Bancroft JD, Layton C. The Hematoxylins and Eosin. In: Suvarna SK, Layton C, Bancroft JD, eds. Bancroft’s Theory and Practice of Histological Techniques. 7th ed. Oxford: Elsevier; 2013 [cited 15 Sept 2025]. p. 173-186. Available in: https://goo.su/2IZsU DOI: https://doi.org/10.1016/B978-0-7020-4226-3.00010-X
Placer ZA, Cushman L, Johnson BC. Estimation of Products of Lipid Peroxidation (malonyl dialdehyde) in Biochemical Systems. Anal. Biochem. [Internet]. 1966; 16(2):359-364. doi: https://doi.org/b96rpj DOI: https://doi.org/10.1016/0003-2697(66)90167-9
Ellman GL, Courtney KD, Andres V, Featherstone RM. A New and Rapid Colorimetric Determination of Acetylcholinesterase Activity. Biochem. Pharmacol. [Internet]. 1961; 7(2):88-95. doi: https://doi.org/fwdkkz DOI: https://doi.org/10.1016/0006-2952(61)90145-9
Aebi H. Catalase in Vitro. Methods Enzymol. [Internet]. 1984; 105:121-126. doi: https://doi.org/dnf7v9 DOI: https://doi.org/10.1016/S0076-6879(84)05016-3
Beutler E. Red Cell Metabolism: A Manual of Biochemical Methods. 2nd ed. New York: Grune and Stratton; 1975.
Sun Y, Oberly LW, Li Y. Simple Method for Clinical Assay of Superoxide Dismutase. Clin. Chem. [Internet]. 1988 [cited 27 Jul 2025]; 34(3):497-500. Available in: https://goo.su/eygPt DOI: https://doi.org/10.1093/clinchem/34.3.497
Sumbuloglu K, Sumbuloglu V. Biostatistics. 19th ed. Ankara: Hatiboglu Publishing; 2019.
Diniz FR, Maia RCAP, de Andrade LRM, Andrade LN, Chaud MV, da Silva CF, Corrêa CB, de Albuquerque-Junior RLC, da Costa LP, Shin SR, Hassan S, Sánchez-López E, Souto EB, Severino P. Silver Nanoparticles-Composing Alginate/Gelatin Hydrogel İmproves Wound Healing in vivo. Nanomaterials. [Internet]. 2020; 10(2):390. doi: https://doi.org/gn725j DOI: https://doi.org/10.3390/nano10020390
Gethin G, Grocott P, Probst S, Clarke E. Current Practice in the Management of Wound Odour: An International Survey. Int. J. Nurs. Stud. [Internet]. 2014; 51(6):865-874. doi: https://doi.org/f55qxr DOI: https://doi.org/10.1016/j.ijnurstu.2013.10.013
Hayouni EA, Miled K, Boubaker S, Bellasfar Z, Abedrabba M, Iwaski H, Oku H, Matsui T, Limam F, Hamdi M. Hydroalcoholic Extract-Based Ointment from Punica granatum L. Peels with Enhanced In vivo Healing Potential on Dermal Wounds. Phytomedicine. [Internet]. 2011; 18(11):976-984. doi: https://doi.org/c34697 DOI: https://doi.org/10.1016/j.phymed.2011.02.011
Yassue-Cordeiro PH, Zandonai CH, Genesi BP, Lopes PS, Sanchez-Lopez E, Garcia ML, Fernandes-Machado NRC, Severino P, Souto EB, Ferreira da Silva C. Development of chitosan/Silver Sulfadiazine/Zeolite Composite Films for Wound Dressing. Pharmaceutics. [Internet]. 2019; 11(10):535. doi: https://doi.org/qsnq DOI: https://doi.org/10.3390/pharmaceutics11100535
do Nascimento MF, Cardoso JC, Santos TS, Tavares LA, Pashirova TN, Severino P, Souto EB, Albuquerque- Junior RLC. Development and Characterization of Biointeractive Gelatin Wound Dressing Based on Extract of Punica granatum L. Pharmaceutics. [Internet]. 2020; 12(12):1204. doi: https://doi.org/qsqw DOI: https://doi.org/10.3390/pharmaceutics12121204
Junker JP, Caterson EJ, Eriksson E. The Microenvironment of Wound Healing. J. Craniofac. Surg. [Internet]. 2013; 24(1):12-16. doi: https://doi.org/f4nxvq DOI: https://doi.org/10.1097/SCS.0b013e31827104fb
Kazemian H, Ghafourian S, Sadeghifard N, Houshmandfar R, Badakhsh B, Taji A, Shavalipour A, Mohebi R, Ebrahim- Saraie HS, Houri H, Heidari H. In vivo Antibacterial and Wound Healing Activities of Roman Chamomile (Chamaemelum Nobile). Infect. Disord. Drug Targets. [Internet]. 2018; 18(1):41-45. doi: https://doi.org/gc77d4 DOI: https://doi.org/10.2174/1871526516666161230123133
Nayak SB, Raju S, Rao VC. Wound Healing Activity of Matricaria recutita L. Extract. J. Wound Care. [Internet]. 2007; 16(7):298-302. doi: https://doi.org/qsq2 DOI: https://doi.org/10.12968/jowc.2007.16.7.27061
Drummond EM, Harbourne N, Marete E, Jacquier J, O’Riordan D, Gibney ER. An In Vivo Study Examining the Anti-İnflammatory Effects of Chamomile, Meadowsweet, and Willow Bark in a Novel Functional Beverage. J. Diet. Suppl. [Internet]. 2013; 10(4):370-380. doi: https://doi.org/qsq4 DOI: https://doi.org/10.3109/19390211.2013.830680
Duman F, Ocsoy I, Kup FO. Chamomile Flower Extract- Directed CuO Nanoparticle Formation for Its Antioxidant and DNA Cleavage Properties. Mater. Sci. Eng. C. [Internet]. 2016; 60:333-338. doi: https://doi.org/f78hgs DOI: https://doi.org/10.1016/j.msec.2015.11.052
Kolodziejczyk-Czepas J, Bijak M, Saluk J, Ponczek MB, Zbikowska HM, Nowak P, Tsirigotis-Maniecka M, Pawlaczyk I. Radical Scavenging and Antioxidant Effects of Matricaria chamomilla Polyphenolic–Polysaccharide Conjugates. Int. J. Biol. Macromol. [Internet]. 2015; 72:1152-1158. doi: https://doi.org/f7ftc7 DOI: https://doi.org/10.1016/j.ijbiomac.2014.09.032
Srivastava JK, Pandey M, Gupta S. Chamomile, a Novel and Selective COX-2 İnhibitor With Anti-Inflammatory Activity. Life Sci. [Internet]. 2009; 85(19-20):663-669. doi: https://doi.org/fjkds3 DOI: https://doi.org/10.1016/j.lfs.2009.09.007
Rummun N, Somanah J, Ramsaha S, Bahorun T, Neergheen-Bhujun VS. Bioactivity of Nonedible Parts of Punica granatum L.: A Potential Source of Functional Ingredients. Int. J. Food Sci. [Internet]. 2013; 2013:602312. doi: https://doi.org/gb64kz DOI: https://doi.org/10.1155/2013/602312
Rahimi HR, Arastoo M, Ostad SN. A comprehensive review of Punica granatum (pomegranate) properties in toxicological, pharmacological, cellular, and molecular biology researches. Iran J. Pharm. Res. [Internet]. 2012 [cited 13 Jul 2025]; 11(2):385-400. Available in: https://goo.su/wafAr
Jarrahi M. An Experimental Study of the Effects of Matricaria chamomilla Extract on Cutaneous Burn Wound Healing in Albino Rats. Nat. Prod. Res. [Internet]. 2008; 22(5):422-427. doi: https://doi.org/d8nrph DOI: https://doi.org/10.1080/14786410701591713
Lukiswanto BS, Miranti A, Sudjarwo SA, Primarizky H, Yuniarti WM. Evaluation of Wound Healing Potential of Pomegranate (Punica granatum) Whole Fruit Extract on Skin Burn Wound in Rats (Rattus norvegicus). J. Adv. Vet. Anim. Res. [Internet]. 2019; 6(2):202-207. doi: https://doi.org/gk7sv4 DOI: https://doi.org/10.5455/javar.2019.f333