Histopathological examination of the effectiveness of nonvascularized tibia and femur bone allografts on bone healing in rat tibia fractures
Abstract
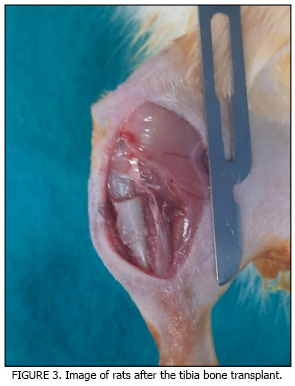
The aim of this study was to histologically evaluate the healing processes of tibia fractures reconstructed with allogeneic bone grafts taken from different anatomical regions (tibia and femur). Twenty-five Sprague-Dawley rats were used in the study. The rats were divided into four groups: fracture control (n = 7), tibia allogeneic bone transplant (n = 7), femur allogeneic bone transplant (n = 7), and donor group (n= 4). Corticocancellous bone blocks (5×5 mm) taken from the donor animals were fixed to the fracture sites created in the tibias of recipient rats with Kirschner wires. At the end of the 4-week healing period, the rats were sacrificed, and the tibias were examined histologically. Tissue samples were fixed in 10 % neutral formalin and decalcified, then embedded in paraffin and stained with hematoxylin-eosin. The new bone formation rate was assessed histomorphometrically and analyzed using the Kruskal–Wallis test. Callus formation was observed at the fracture line in all groups. Callus tissue was more uniform and new bone formation was more dense in the tibia and femur allogeneic transplant groups. The highest new bone formation was observed in the tibia and femur allogeneic bone transplant group when compared with controls (P< 0,05 P = 0.002). Non vascular allogeneic bone transplantation; tibia and femur derived grafts, was found to significantly increase fracture healing compared to the control group. It was concluded that the use of allogeneic bone obtained from different anatomic sites may be an effective and biocompatible option for bone regeneration.
Downloads
References
Lelo I, Calabrese G, De Luca G, Conoci S. Recent Advances in Hydroxyapatite-Based Biocomposites for Bone Tissue Regeneration in Orthopedics. Int. J. Mol. Sci. [Internet]. 2022; 23(17):9721. doi: https://doi.org/g8rzn6 DOI: https://doi.org/10.3390/ijms23179721
Sun J, Xie W, Wu Y, Li Z, Li Y. Accelerated Bone Healing via Electrical Stimulation. Adv. Sci. [Internet]. 2025; 12(24):e2404190. doi: https://doi.org/g8694w DOI: https://doi.org/10.1002/advs.202404190
Omi M, Mishina Y. Roles of osteoclasts in alveolar bone remodeling. Genesis. [Internet]. 2022; 60(8-9):e23490. doi: https://doi.org/gtz76m DOI: https://doi.org/10.1002/dvg.23490
Maruyama M, Rhee C, Utsunomiya T, Zhang N, Ueno M, Yao Z, Goodman SB. Modulation of the inflammatory response and bone healing. Front. Endocrinol. [Internet]. 2020; 11:386. doi: https://doi.org/gjtsz6 DOI: https://doi.org/10.3389/fendo.2020.00386
Moriarty TF, Metsemakers WJ, Morgenstern M, Hofstee MI, Vallejo-Diaz A, Cassat JE, Wildemann B, Depypere M, Schwarz EM, Richards RG. Fracture-related infection. Nat. Rev. Dis. Primers. [Internet]. 2022; 8(1):67. doi: https://doi.org/mq9t DOI: https://doi.org/10.1038/s41572-022-00396-0
Steppe L, Megafu M, Tschaffon-Müller ME, Ignatius A, Haffner-Luntzer M. Fracture healing research: Recent insights. Bone Rep. [Internet]. 2023; 19:101686. doi: https://doi.org/g4nz6p DOI: https://doi.org/10.1016/j.bonr.2023.101686
Shu LZ, Zhang XL, Ding YD, Lin H. From inflammation to bone formation: the intricate role of neutrophils in skeletal muscle injury and traumatic heterotopic ossification. Exp. Mol. Med. [Internet]. 2024; 56(7):1523-30. doi: https://doi.org/g8xfdh DOI: https://doi.org/10.1038/s12276-024-01270-7
Camarena A, Kang L, Mirando AJ, Augustine E, McMillian NS, Stinson NC, Agarwal SM, Becker ML, Hilton MJ, Fernandez-Moure JS. Platelet-rich plasma enhances rib fracture strength and callus formation in vivo. J. Trauma Acute Care Surg. [Internet]. 2024; 97(6):884-890. doi: https://doi.org/qmx3 DOI: https://doi.org/10.1097/TA.0000000000004441
Trompet D, Melis S, Chagin AS, Maes C. Skeletal stem and progenitor cells in bone development and repair. J. Bone Miner. Res. [Internet]. 2024; 39(6):633-654. doi: https://doi.org/gt2h8d DOI: https://doi.org/10.1093/jbmr/zjae069
Hente RW, Perren SM. Tissue deformation controlling fracture healing. J. Biomech. [Internet]. 2021; 125:110576. doi: https://doi.org/qmx4 DOI: https://doi.org/10.1016/j.jbiomech.2021.110576
Wang YH, Zhao CZ, Wang RY, Du QX, Liu JY, Pan J. The crosstalk between macrophages and bone marrow mesenchymal stem cells in bone healing. Stem Cell Res. Ther. 2022; 13(1):524. doi: https://doi.org/qscv DOI: https://doi.org/10.1186/s13287-022-03211-5
ZhaoQ, Liu X, Yu C, Xiao Y. Macrophages and Bone Marrow-Derived Mesenchymal Stem Cells Work in Concert to Promote Fracture Healing: A Brief Review. DNA Cell Biol. [Internet]. 2022; 41(3):276-284. doi: https://doi.org/grs8x6 DOI: https://doi.org/10.1089/dna.2021.0869
Rodríguez-Merchán EC. A Review of Recent Developments in the Molecular Mechanisms of Bone Healing. Int. J. Mol. Sci. [Internet]. 2021; 22(2):767. doi: https://doi.org/gscc6q DOI: https://doi.org/10.3390/ijms22020767
Bingül MB, Gül M, Dundar S, Tanik A, Artas G, Polat ME. Enhanced Bone Healing Through Systemic Capsaicin Administration: An Experimental Study on Wistar Rats. Med. Sci. Monit. [Internet]. 2024; 30:942485. doi: https://doi.org/qscw DOI: https://doi.org/10.12659/MSM.942485
Miron RJ. Optimized bone grafting. Periodontol. 2000. [Internet]. 2024; 94(1):143-160. doi: https://doi.org/g9dx5s DOI: https://doi.org/10.1111/prd.12517
Sanz-Sánchez I, Sanz-Martín I, Ortiz-Vigón A, Molina A, Sanz M. Complications in bone-grafting procedures: Classification and management. Periodontol. 2000. [Internet]. 2022; 88(1):86-102. doi: https://doi.org/gts4dr DOI: https://doi.org/10.1111/prd.12413
Schmidt AH. Autologous bone graft: Is it still the gold standard? Injury. [Internet]. 2021; 52(2):18-22. doi: https://doi.org/gnn7qp DOI: https://doi.org/10.1016/j.injury.2021.01.043
Zhao R, Yang R, Cooper PR, Khurshid Z, Shavandi A, Ratnayake J. Bone Grafts and Substitutes in Dentistry: A Review of Current Trends and Developments. Molecules. [Internet]. 2021; 26(10):3007. doi: https://doi.org/gsmsrh DOI: https://doi.org/10.3390/molecules26103007
Goutam M, Batra N, Jyothirmayee K, Bagrecha N, Deshmukh P, Malik S. A comparison of xenograft graft material and synthetic bioactive glass allograft in immediate dental implant patients. J. Pharm. Bioallied Sci. [Internet]. 2022; 14(1):980-982. doi: https://doi.org/qscz DOI: https://doi.org/10.4103/jpbs.jpbs_808_21
Ferraz MP. Bone Grafts in Dental Medicine: An Overview of Autografts, Allografts and Synthetic Materials. Materials. [Internet]. 2023; 16(11):4117. doi: https://doi.org/ DOI: https://doi.org/10.3390/ma16114117
Lee SS, Huber S, Ferguson SJ. Comprehensive in vitro comparison of cellular and osteogenic response to alternative biomaterials for spinal implants. Mater. Sci. Eng. C. [Internet]. 2021; 127:112251. doi: https://doi.org/qsc2 DOI: https://doi.org/10.1016/j.msec.2021.112251
Stopa Z, Siewert-Gutowska M, Abed K, Szubinska-Lelonkiewicz D, Kaminski A, Fiedor P. Evaluation of the safety and clinical efficacy of allogeneic bone grafts in the reconstruction of the maxilla and mandible. Transplant. Proc. [Internet]. 2018; 50(7):2199-2201. doi: https://doi.org/gfchtk DOI: https://doi.org/10.1016/j.transproceed.2018.02.122
Ippolito JA, Martinez M, Thomson JE, Willis AR, Beebe KS, Patterson FR, Benevenia J. Complications following allograft reconstruction for primary bone tumors: Considerations for management. J. Orthop. [Internet]. 2019; 16(1):49-54. doi: https://doi.org/gj33n7 DOI: https://doi.org/10.1016/j.jor.2018.12.013
Zou W, Li X, Li N, Guo T, Cai Y, Yang X, Liang J, Sun Y, Fan Y. A comparative study of autogenous, allograft and artificial bone substitutes on bone regeneration and immunotoxicity in rat femur defect model. Regen. Biomater. [Internet]. 2020; 8(1):40. doi: https://doi.org/qsc9 DOI: https://doi.org/10.1093/rb/rbaa040
Ciszynski M, Dominiak S, Dominiak M, Gedrange T, Hadzik J. Allogenic Bone Graft in Dentistry: A Review of Current Trends and Developments. Int. J. Mol. Sci. [Internet]. 2023; 24(23):16598. doi: https://doi.org/qqcn DOI: https://doi.org/10.3390/ijms242316598
Baldwin P, Li DJ, Auston DA, Mir HS, Yoon RS, Koval KJ. Autograft, Allograft, and Bone Graft Substitutes: Clinical Evidence and Indications for Use in the Setting of Orthopaedic Trauma Surgery. J. Orthop. Trauma. [Internet]. 2019; 33(4):203-213. doi: https://doi.org/gmtzbb DOI: https://doi.org/10.1097/BOT.0000000000001420
Sheen JR, Mabrouk A, Garla VV. Fracture healing overview. In: StatPearls. [internet]. Treasure Island, FL: StatPearls Publishing. 2023 [20 Aug 2025]. Available in: https://goo.su/qx3Z
Pajarinen J, Lin T, Gibon E, Kohno Y, Maruyama M, Nathan K, Lu L, Yao Z, Goodman SB. Mesenchymal stem cellmacrophage crosstalk and bone healing. Biomaterials. [Internet]. 2019; 196:80-89. doi: https://doi.org/gh4vp4 DOI: https://doi.org/10.1016/j.biomaterials.2017.12.025
Tanrisever M, Tekin B, Can UK, Istek O, Ozcan EC, Ozercan IH, Gelic T, Dundar S. The Effect of Local Melatonin Application on Bone Fracture Healing in Rat Tibias. Medicina. [Internet]. 2025; 61(1):146. doi: https://doi.org/qmzf DOI: https://doi.org/10.3390/medicina61010146
Ciobanu P, Danciu M, Pascu A, Gardikiotis I, Forna N, Sirbu MT, Calistru AE, Puha B, Veliceasa B, Sirbu PD. Experimental Study on Rats with Critical-Size Bone Defects Comparing Effects of Autologous Bone Graft, Equine Bone Substitute Bio-Gen® Alone or in Association with Platelet-Rich Fibrin (PRF). Polymers. [Internet]. 2024; 16(11):1502. doi: https://doi.org/qsdb DOI: https://doi.org/10.3390/polym16111502
Vorontsov P, Maltseva VY. A review of animal models for bone fracture nonunion and their role in studying biological therapy efficacy. Orthop. Traumatol. Prosthet. [Internet]. 2024; 2:81–87. doi: https://doi.org/qsdc DOI: https://doi.org/10.15674/0030-59872024281-87
Marongiu G, Contini A, Cozzi Lepri A, Donadu M, Verona M, Capone A. The Treatment of Acute Diaphyseal Longbones Fractures with Orthobiologics and Pharmacological Interventions for Bone Healing Enhancement: A Systematic Review of Clinical Evidence. Bioengineering. [Internet]. 2020; 7(1):22. doi: https://doi.org/qsdd DOI: https://doi.org/10.3390/bioengineering7010022
Meiser S, Arora R, Petersen J, Keiler A, Liebensteiner MC, Pallua JD, Wurm A. Radiographic and clinical outcome of tibial plateau fractures treated with bone allograft. Arch. Orthop. Trauma Surg. [Internet]. 2023; 143(5):24472454. doi: https://doi.org/qsdf DOI: https://doi.org/10.1007/s00402-022-04461-x
Bahadoran Z, Mirmiran P, Kashfi K, Ghasemi A. Importance of Systematic Reviews and Meta-analyses of Animal Studies: Challenges for Animal-to-Human Translation. J. Am. Assoc. Lab. Anim. Sci. [Internet]. 2020; 59(5):469-477. doi: https://doi.org/gg637h DOI: https://doi.org/10.30802/AALAS-JAALAS-19-000139
Durmaz B, Gunes N, Koparal M, Gul M, Dundar S, Bingul MB. Investigation of the effects of quercetin and xenograft on the healing of bone defects: An experimental study. J. Oral Biol. Craniofac. Res. [Internet]. 2023; 13(1):22-27. doi: https://doi.org/pmp9 DOI: https://doi.org/10.1016/j.jobcr.2022.10.008