Anomalía gloso-mandibular congénita con hipoplasia mandibular severa en un cerdo. Reporte de caso
Resumen
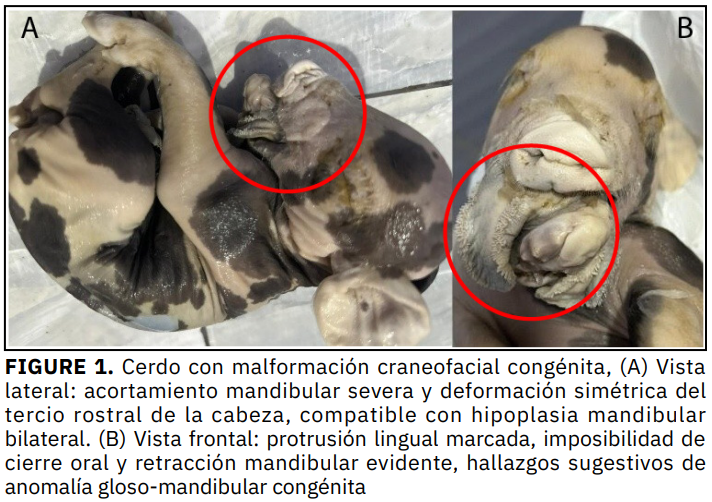
Las malformaciones congénitas en animales afectan la producción, debido a su impacto en la sobrevivencia neonatal, el bienestar del animal y la eficiencia productiva. Se describe un caso patológico en un cerdo recién nacido, proveniente de una crianza familiar, distrito de Baños del Inca, Cajamarca (Perú). El espécimen fue examinado en el Laboratorio de Patología Veterinaria de la Universidad Nacional de Cajamarca, donde se realizó una evaluación morfológica y radiográfica. Las radiografías laterolateral y dorsoventral evidenciaron una hipoplasia mandibular bilateral severa, con desplazamiento dorsal de la sínfisis y acortamiento simétrico de ambas ramas mandibulares. Los hallazgos fueron compatibles anomalía gloso-mandibular congénita con hipoplasia mandibular severa, contribuyendo al conocimiento de las malformaciones craneofaciales en especies domésticas, enfatizando la importancia del diagnóstico morfológico y radiográfico en neonatos. El presente trabajo tuvo como objetivo describir un caso patológico en un cerdo.
Descargas
Citas
Larsen WJ. Embriología humana y biología del desarrollo. 5ª ed. Elsevier; 2018.
Tucker B, Craig J, Morrison R, Smith R, Kirkwood. Piglet Viability: A Review of Identification and Pre-Weaning Management Strategies. Animals. [Internet]. 2021; 11(10):2902. doi: https://doi.org/q4pv DOI: https://doi.org/10.3390/ani11102902
Pourlis A, Papakonstantinou GI, Doukas D, Papatsiros VG. Overview of Swine Congenital Malformations Associated with Abnormal Twinning. Vet. Sci. [Internet]. 2023; 10(9):534. doi: https://doi.org/q4pw DOI: https://doi.org/10.3390/vetsci10090534
Couly G, Creuzet S, Bennaceur S, Vincent C, Le Douarin NM. Interactions between Hox-negative cephalic neural crest cells and the foregut endoderm in patterning the facial skeleton in the vertebrate head. Development. [Internet]. 2002; 129(4):1061–1073. doi: https://doi.org/q4p2 DOI: https://doi.org/10.1242/dev.129.4.1061
Funato N, Kokubo H, Nakamura M, Yanagisawa H, Saga Y. Specification of jaw identity by the Hand2 transcription factor. Sci. Rep. [Internet]. 2016; 6:28405. doi: https://doi.org/f8s55c DOI: https://doi.org/10.1038/srep28405
Barron F, Woods C, Kuhn K, Bishop J, Howard MJ, Clouthier DE. Downregulation of Dlx5 and Dlx6 expression by Hand2 is essential for initiation of tongue morphogenesis. Development. [Internet]. 2011; 138(11):2249–2259. doi: https://doi.org/ckksw6 DOI: https://doi.org/10.1242/dev.056929
Schmotzer C, Shehata B. Two Cases of Agnathia (Otocephaly): With Review of the Role of Fibroblast Growth Factor (FGF8) and Bone Morphogenetic Protein (BMP4) in Patterning of the First Branchial Arch. Pediatr. Dev. Pathol. [Internet]. 2008; 11(4):321-324. doi: https://doi.org/fkw88f DOI: https://doi.org/10.2350/07-09-0351.1
Grevellec A, Tucker AS. The pharyngeal pouches and clefts: development, evolution, structure and derivatives. Semin. Cell Dev. Biol. [Internet]. 2010; 21(3):325–332. doi: https://doi.org/dx262z DOI: https://doi.org/10.1016/j.semcdb.2010.01.022
Ruest LB, Xiang X, Lim KC, Levi G, Clouthier DE. Endothelin-A receptor-dependent and -independent signaling pathways in establishing mandibular identity. Development. [Internet]. 2004; 131(18):4413–4423. doi: https://doi.org/dcn2bz DOI: https://doi.org/10.1242/dev.01291
Depew MJ, Lufkin T, Rubenstein JL. Specification of jaw subdivisions by Dlx genes. Science. [Internet]. 2002; 298(5592):381–385. doi: https://doi.org/cbrj4 DOI: https://doi.org/10.1126/science.1075703
Handrigan GR, Richman JM. Unicuspid and bicuspid tooth crown formation in squamates. J. Exp. Zool. B Mol. Dev. Evol. [Internet]. 2011; 316B(8):598–608. doi: https://doi.org/ckt6mw DOI: https://doi.org/10.1002/jez.b.21438
Braybrook C, Doudney K, Marçano AC, Arnason A, Bjornsson A, Patton MA, Goodfellow PJ, Moore GE, Stanier P. The T-box transcription factor gene TBX22 is mutated in X-linked cleft palate and ankyloglossia. Nat. Genet. [Internet]. 2001; 29(2):179–183. doi: https://doi.org/c2ttw3 DOI: https://doi.org/10.1038/ng730
Bille N, Nielsen NC. Congenital malformations in pigs in a post mortem material. Nord Vet. Med. 1977 [cited 20 Dec 2025]; 29:128-136. PMID: 870881. Available in: https://goo.su/e9XUY3B
Medeiros DM, Crump JG. New perspectives on pharyngeal dorsoventral patterning in development and evolution of the vertebrate jaw. Dev. Biol. [Internet]. 2012; 372(2):121-135. doi: https://doi.org/f4cg7d DOI: https://doi.org/10.1016/j.ydbio.2012.08.026
Minoux M, Rijli FM. Molecular mechanisms of cranial neural crest cell migration and patterning in craniofacial development. Development. [Internet]. 2010; 137(16):2605–2621. doi: https://doi.org/d33fv5 DOI: https://doi.org/10.1242/dev.040048
Depew MJ, Simpson CA. 21st century neontology and the comparative development of the vertebrate skull. Dev Dyn. [Internet]. 2006; 235(5):1256-1291. doi: https://doi.org/d97dxp DOI: https://doi.org/10.1002/dvdy.20796
Bildsoe H, Loebel DA, Jones VJ, Hor ACC, Braithwaite AW, Chen YT, Behringer RR, Tam PPL. The mesenchymal architecture of the cranial mesoderm of mouse embryos is disrupted by the loss of Twist1 function. Dev. Biol. [Internet]. 2013; 374(2):295–307. doi: https://doi.org/f4pp6q DOI: https://doi.org/10.1016/j.ydbio.2012.12.004
Barriga EH, Trainor PA, Bronner M, Mayor R. Animal models for studying neural crest development: is the mouse different? Development. [Internet]. 2015; 142(9):1555–1560. doi: https://doi.org/f69xpn DOI: https://doi.org/10.1242/dev.121590
Trainor PA. Neural crest cells: evolution, development and disease. London: Academic Press; 2014 [cited 20 Dec 2025]. Available: https://goo.su/osPAQxX
Bronner ME, LeDouarin NM. Development and evolution of the neural crest: an overview. Dev. Biol. [Internet]. 2012; 366(1):2–9. doi: https://doi.org/fxpjmj DOI: https://doi.org/10.1016/j.ydbio.2011.12.042